Methylnitronitrosoguanidine
| |||||||||||||||||||||||||||||||||||||||||||||||||
Read other articles:

Kentucky Indy 300IndyCar SeriesTempatKentucky SpeedwayLomba pertama2000Lomba terakhir2011Jarak tempuh300 miles (483 km)Jumlah putaran200Nama sebelumnyaBelterra Casino Indy 300 (2000–2004)AMBER Alert Portal Indy 300 (2005)Meijer Indy 300 presented by Coca-Cola and Secret (2006)Meijer Indy 300 presented by Coca-Cola and Edy's (2007–2009)Kentucky Indy 300 (2010–2011)Terbanyak menang(pengemudi)Buddy Lazier (2)Sam Hornish Jr. (2)Terbanyak menang(tim)Penske Racing (3) Kentucky Indy 300 adalah ba…

Mesin KR Toyota adalah seri mesin piston 3 segaris yang didesain oleh Daihatsu, anak perusahaan Toyota. Seri 1KR menggunakan blok mesin dari aluminium dan kepala silinder DOHC. Telah menggunakan injeksi multi-point dengan teknologi VVT-i. Bobot mesin ini pun hanya 69 kg dan dipakai di mobil kota. Artikel ini membutuhkan rujukan tambahan agar kualitasnya dapat dipastikan. Mohon bantu kami mengembangkan artikel ini dengan cara menambahkan rujukan ke sumber tepercaya. Pernyataan tak bersumber bisa …

Hadromerida Tethya aurantia Status konservasi Data Kurang (IUCN 2.3) Klasifikasi ilmiah Kerajaan: Animalia Filum: Porifera Kelas: Demospongiae Ordo: HadromeridaTopsent, 1894 Families Acanthochaetetidae Fischer, 1970 Alectonidae Rosell, 1996 Choanitidae Clionaidae (replacement name for Clionidae d'Orbigny, 1851) Hemiasterellidae Lendenfeld, 1889 Placospongiidae Gray, 1867 Polymastiidae Gray, 1867 Sollasellidae Lendenfeld, 1887 Spirastrellidae Ridley & Dendy, 1886 Stylocordylidae Topsent,…

Cet article est une ébauche concernant le Concours Eurovision de la chanson et l’Arménie. Vous pouvez partager vos connaissances en l’améliorant (comment ?) ; pour plus d’indications, visitez le projet Eurovision. Arménieau Concours Eurovision 2010 Données clés Pays Arménie Chanson Apricot Stone Interprète Eva Rivas Compositeur Armen Martirosyan Parolier Karen Kavaleryan Langue Anglais Sélection nationale Type de sélection jury + télévote Date 14 février 2010 C…

Abu UrwahLahirUrwahc. 1983Meninggal17 Agustus 2015Poso Pesisir Utara, PosoSebab meninggalDitembak matiKebangsaanIndonesiaNama lainBado, OsamaOrganisasiMujahidin Indonesia TimurDikenal atasTerorisme, PengebomanGelarAnggota Mujahidin Indonesia TimurLawan politikKepolisian Negara Republik IndonesiaTentara Nasional IndonesiaSuami/istriNurifa Abu Urwah alias Bado atau Osama (lahir c. 1983 - meninggal 17 Agustus 2015 di Poso Pesisir Utara, Poso), adalah seorang militan Islam Indo…

Teodorus dari AmaseaMartirLahirtidak diketahuiEukhaita (?) atau Alasium, Turki Greatmartyr Theodore the Tyro (“the Recruit”) - Orthodox Church in AmericaMeninggal17 Februari 306Amasea, TurkiPestagereja katolik roma: 9 Novembergereja ortodoks:17 Februari dan Sabtu pertama di Agung Prapaskah;Korcula: 27 JuliAtributBerpakaian sebagai tentara terkadang berpakaian pengadilan, dengan emblem seperti tombak, bait, obor, naga, kayu bakar, dan karangan bunga martirPelindungBrindisi, pemulihan artikel …

PT DahanaJenisPerseroan terbatasIndustriBahan peledakDidirikan22 Oktober 1966KantorpusatSubang, IndonesiaWilayah operasiIndonesiaTokohkunciWildan Widarman[1](Direktur Utama)Donny Ermawan Taufanto[2](Komisaris Utama)ProdukBahan peledakMerekDayagelDayadetDANFODABEXJasaPengeboran & peledakanPenghancuran bangunanPemusnahan bahan peledakPengangkutan bahan peledakPenyimpanan bahan peledakPendapatanRp 3,322 triliun (2022)[3]Laba bersihRp 260,7 milyar (2022)[3]Total a…

U.S. House district for Florida Florida's 7th congressional districtInteractive map of district boundaries since January 3, 2023Representative Cory MillsR–New Smyrna BeachArea436[1] sq mi (1,130 km2)Distribution97.87% urban[2]2.13% ruralPopulation (2022)788,222[3]Median householdincome$75,437[3]Ethnicity61.5% White20.8% Hispanic8.8% Black4.2% Two or more races3.9% Asian0.8% otherCook PVIR+5[4] Florida's 7th congressional district is …

Pour les articles homonymes, voir Arfi. Fabrice ArfiFabrice Arfi en 2018BiographieNaissance 4 septembre 1981 (42 ans)LyonNationalité françaiseActivité Journaliste d'investigationRédacteur à MediapartAutres informationsA travaillé pour Le MondeMediapartLe ParisienLibérationMembre de Consortium international des journalistes d'investigationDistinction Prix éthique d'Anticor (2015)modifier - modifier le code - modifier Wikidata Fabrice Arfi, né le 4 septembre 1981 à Lyon, est un jour…

Gugus Quintuplet. Gugus Quintuplet adalah gugus terbuka padat yang terletak pada jarak 26.000 tahun cahaya dari Bumi ke arah konstelasi Sagitarius dan hanya 100 tahun cahaya dari pusat galaksi. Gugus Quintuplet awalnya terkenal karena lima bintangnya yang sangat terkenal, tetapi sekarang diketahui mengandung banyak bintang bercahaya masif yang tidak terdeteksi gelombang yang terlihat karena debu di sepanjang garis pandang. Memiliki massa yang setara lebih dari 10.000 bintang seperti matahari kit…

В Википедии есть статьи о других людях с фамилиями Каменев и Розенфельд. Лев Борисович КаменевЛев Розенфельд член Политбюро ЦК РКП(б) и председатель СТО Л.Б. Каменев Председатель Всероссийского ЦИК 27 октября (9 ноября) — 8 (21) ноября 1917 Глава правительства Владимир Ленин П…

Keuskupan LucenaDioecesis LucenensisDiyosesis ng LucenaKatolik Lambang Keuskupan LucenaLokasiNegara FilipinaWilayahbagian tengah Provinsi QuezonProvinsi gerejawiLipaKoordinat13°56′9.4″N 121°36′45″E / 13.935944°N 121.61250°E / 13.935944; 121.61250Koordinat: 13°56′9.4″N 121°36′45″E / 13.935944°N 121.61250°E / 13.935944; 121.61250StatistikLuas2.334 km2 (901 sq mi)Populasi- Total- Katolik(per 2…

District of Telangana in IndiaMahabubabad districtDistrict of TelanganaEpiphany Cathedral, DornakalLocation in TelanganaMahabubabad districtCountryIndiaStateTelanganaHeadquartersMahabubabadMandalas16Government • District collectorSri V.P.Goutham • Parliament constituenciesMahabubabad • Assembly constituenciesMahabubabad, DornakalArea • Total2,876.70 km2 (1,110.70 sq mi)Population (2011) • Total774,549 • D…

ХристианствоБиблия Ветхий Завет Новый Завет Евангелие Десять заповедей Нагорная проповедь Апокрифы Бог, Троица Бог Отец Иисус Христос Святой Дух История христианства Апостолы Хронология христианства Раннее христианство Гностическое христианство Вселенские соборы Ни�…

Andai Ia TahuSutradaraIndra YudhistiraProduserWishnutamaDitulis olehMonty TiwaIndra YudhistiraPemeranRachel MaryamMarcell SiahaanNadia MulyaMelanie SadonoPriscilla CameliaHarry PancaAlex AbbadPenata musikAndi RiantoPenyuntingFredy AryantoRamdan PanigoroDistributorTransinema PicturesTanggal rilis19 Desember 2002Durasi99 menitNegaraIndonesia Andai Ia Tahu adalah sebuah film yang disutradarai oleh Indra Yudhistira dan diproduseri oleh Trans TV. Film ini dirilis pada 19 Desember 2002 di seluru…
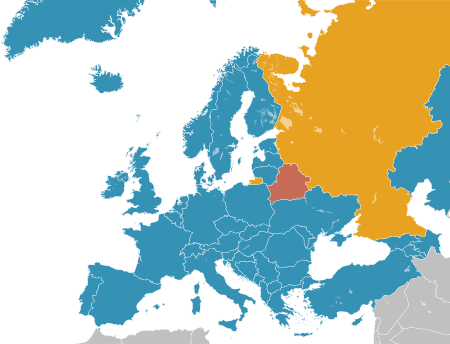
Overview of the state of capital punishment in Belgium Europe holds the greatest concentration of abolitionist states (blue). Map current as of 2022 Abolished for all offences Abolished in practice Retains capital punishment Capital punishment in Belgium was formally abolished on August 1, 1996, for all crimes, in both peacetime and wartime.[1] The last execution for crimes committed in peacetime took place in July 1863, when in Ypres a farmer was exec…

American sportscaster Dan DickersonDickerson at Comerica Park in 2011BornDaniel Hill Dickerson (1958-11-13) November 13, 1958 (age 65)NationalityAmericanEducationOhio Wesleyan University (B.A.)Sports commentary careerTeamDetroit Tigers (2000–present)GenrePlay-by-playSportMajor League Baseball Daniel Hill Dickerson (born November 13, 1958)[1] is an American sportscaster, best known for his current position as the lead radio play-by-play voice of Major League Baseball's Detroit Tige…

Group of rock formations within the Cape Supergroup sequence of rocks Main article: Cape Fold Belt § Cape Supergroup Table Mountain seen from Signal Hill, across the Cape Town city bowl. The portion of the mountain made up of Table Mountain Sandstone is indicated on the right.[1] It is this mountain that has given its name to the geological structure that occurs in the mountains throughout the Western Cape Schematic diagram of an approximate 100 km west-east (left to right) ge…

هنودمعلومات عامةنسبة التسمية الهند التعداد الكليالتعداد قرابة 1.21 مليار[1][2]تعداد الهند عام 2011ق. 1.32 مليار[3]تقديرات عام 2017ق. 30.8 مليون[4]مناطق الوجود المميزةبلد الأصل الهند البلد الهند الهند نيبال 4,000,000[5] الولايات المتحدة 3,982,398[6] الإمارا�…

Pour les articles homonymes, voir Latour. Bruno LatourBruno Latour en 2017.FonctionsDirecteur du Programme d'expérimentation en arts politiquesjuin 2012 - août 2017Directeur ou directrice scientifique (en)Institut d'études politiques de Paris2007-2012Christine MusselinBiographieNaissance 22 juin 1947Beaune (Côte-d'Or, Bourgogne-Franche-Comté, France)Décès 8 octobre 2022 (à 75 ans)13e arrondissement de Paris (Paris, Île-de-France, France)Nom de naissance Bruno Paul Louis LatourNatio…

